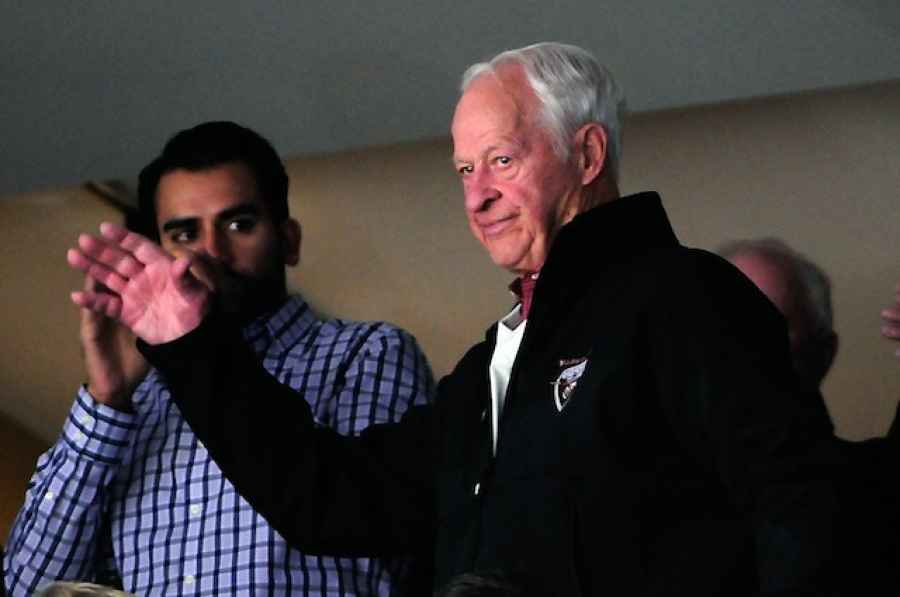

22 Dec 2014 : 12:25PM
After suffering a major stroke in late October, the health of Detroit Red Wings legend Gordie Howe seemed to be rapidly deteriorating.
But lately there have been reports that his condition has been steadily improving to the point where he is now able to help with household chores, reports that were confirmed by his family on Friday. That is his when his family released a statement through the Red Wings organization thanking an experimental stem cell treatment for what they describe as a "miraculous" recovery.
Here is the complete statement from the Howe family.
"Following the press coverage of our father's deteriorating medical condition, the Howe Family was contacted in late November by Dr. Maynard Howe (CEO) and Dave McGuigan (VP) of Stemedica Cell Technologies. McGuigan knew our family as a result of his previous employment with the Detroit Red Wings. Stemedica is a biotechnology company that manufactures allogeneic adult stem cells in its U.S. government licensed, cGMP facility in San Diego, California. Although no relation, Dr. Howe and his brothers Drs. David and Roger are hockey players and big Gordie Howe fans, having grown up in Minnesota. They wished to help our father by generously facilitating Dad's participation in a stem cell clinical trial at Novastem, a licensed distributor of Stemedica's products in Mexico.
Novastem ([link]
But lately there have been reports that his condition has been steadily improving to the point where he is now able to help with household chores, reports that were confirmed by his family on Friday. That is his when his family released a statement through the Red Wings organization thanking an experimental stem cell treatment for what they describe as a "miraculous" recovery.
Here is the complete statement from the Howe family.
"Following the press coverage of our father's deteriorating medical condition, the Howe Family was contacted in late November by Dr. Maynard Howe (CEO) and Dave McGuigan (VP) of Stemedica Cell Technologies. McGuigan knew our family as a result of his previous employment with the Detroit Red Wings. Stemedica is a biotechnology company that manufactures allogeneic adult stem cells in its U.S. government licensed, cGMP facility in San Diego, California. Although no relation, Dr. Howe and his brothers Drs. David and Roger are hockey players and big Gordie Howe fans, having grown up in Minnesota. They wished to help our father by generously facilitating Dad's participation in a stem cell clinical trial at Novastem, a licensed distributor of Stemedica's products in Mexico.
Novastem ([link]
) is currently conducting federally licensed and Institutional Review Board approved clinical trials for several medical conditions, including stroke, using Stemedica's stem cell products. At the time we were contacted, Mr. Hockey had been rapidly declining and was essentially bedridden with little ability to communicate or to eat on his own.
After reviewing the information on Stemedica and Novastem, our family decided to give our father this opportunity. On December 8, Mr. Hockey underwent a two-day, non-surgical treatment at Novastem's medical facility. The treatment included neural stem cells injected into the spinal canal on Day 1 and mesenchymal stem cells by intravenous infusion on Day 2. His response was truly miraculous. At the end of Day 1 he was walking with minimal effort for the first time since his stroke. By Day 2 he was conversing comfortably with family and staff at the clinic.
On the third day, he walked to his seat on the plane under his own power. By Day 5 he was walking unaided and taking part in helping out with daily household chores. When tested, his ability to name items has gone from less than 25 percent before the procedure to 85 percent today. His physical therapists have been astonished. Although his short-term memory, strength, endurance and coordination have plenty of room for improvement, we are hopeful that he will continue to improve in the months to come.
As a family, we are thrilled that Dad's quality of life has greatly improved, and his progress has exceeded our greatest expectations. Once again, we cannot emphasize how much you have fueled Mr. Hockey's recovery and we thank everyone for their continued prayers and support."
After reviewing the information on Stemedica and Novastem, our family decided to give our father this opportunity. On December 8, Mr. Hockey underwent a two-day, non-surgical treatment at Novastem's medical facility. The treatment included neural stem cells injected into the spinal canal on Day 1 and mesenchymal stem cells by intravenous infusion on Day 2. His response was truly miraculous. At the end of Day 1 he was walking with minimal effort for the first time since his stroke. By Day 2 he was conversing comfortably with family and staff at the clinic.
On the third day, he walked to his seat on the plane under his own power. By Day 5 he was walking unaided and taking part in helping out with daily household chores. When tested, his ability to name items has gone from less than 25 percent before the procedure to 85 percent today. His physical therapists have been astonished. Although his short-term memory, strength, endurance and coordination have plenty of room for improvement, we are hopeful that he will continue to improve in the months to come.
As a family, we are thrilled that Dad's quality of life has greatly improved, and his progress has exceeded our greatest expectations. Once again, we cannot emphasize how much you have fueled Mr. Hockey's recovery and we thank everyone for their continued prayers and support."
This news item is from Stemaid™ : Pluripotent Stem Cells
https://stemaid.com/news/gordie-howes-condition-improving-following-stem-cell-treatment.html
Print this page